Imaging the developing brain at Ultra-High Field
Scanning the brain at ultra-high (7 Tesla) field presents significant gains in signal to noise ratio and specific contrasts and thus exciting new opportunities for learning about the developing brain. These include high resolution imaging of key structures such as the developing cortex, higher sensitivity and specificity for functional imaging, improved visualisation of the cerebral vasculature, and increased sensitivity for neurometablites with MRS. By working closely with Dr Shaihan Malik (Physics lead) and Philippa Bridgen (radiography lead), engineering, and clinical colleagues we have established the UK’s first ultra-high field imaging programme on the 7T Siemens system on the London Collaborative Ultra-High Field system (LoCUS) at St Thomas’ Hospital. We have safely scanned infants as young as 33+4 weeks postmenstrual age and weighting as little as 1.6kg.
Our first papers describing this work, the safety and first acquisitions and T1/T2 values for ultra-high field neonatal imaging can be found here:
Mahmoud et al. T1 and T2 measurements of the neonatal brain at 7T. Magn Reson Med 2024; Nov 3
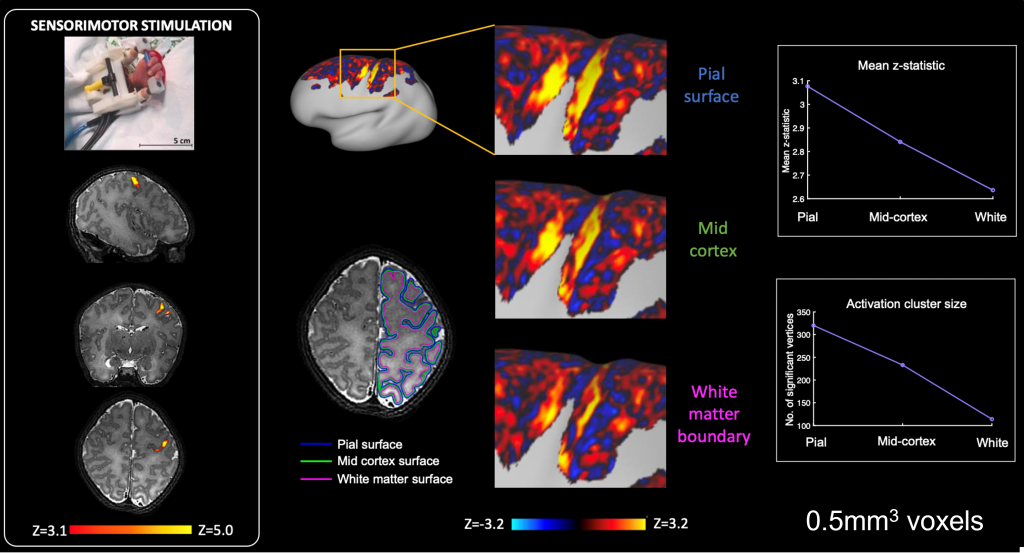
It is known that the sensitivity and spatial specificity of fMRI is greatly enhanced at ultra-high field. This presents unique opportunities to study how brain activity emerges in the neonatal brain and explore the contribution of cortical layers together with the developing vasculature to the BOLD response. In collaboration with Professor Jon Polimeni (Stanford & Harvard MGH) we have recently begun acquiring high resolution ultra-high field fMRI data from neonates.
A pre-print describing the first stages of this work including characterisation of depth specific haemodynamic responses can be found here:
This work was started by funding from Action Medical Research and is currently funded through the Medical Research Council
Simultaneous EEG-fMRI
Working with our collaborators at UCL (led by Dr Lorenzo Fabrizi) we are collecting simultaneous EEG and fMRI data from neonates. We used this methodology to study spontaneous bursting neuronal activity which is a hallmark of the developing brain and thought to be crucial for establishing neural circuits. For the first time in preterm human infants, we identified the source of the most common type of this activity in the late preterm period (posterior temporal delta brushes) as the insula, thus identifying it as a key hub in the developing human brain. This was the first description of simultaneous EEG-fMRI and demonstration of the link between electrical neural activity and the fMRI haemodynamic response in human infancy. The work was recognised by a Merit Abstract Award (Organisation of Human Brain Mapping) and significant media attention (ie, FT, Independent).

Please also refer to the article published in Elife in 2017: https://cdn.elifesciences.org/articles/27814/elife-27814-v1.pdf
This work was funded by a Clinician Scientist Fellowship from the Medical Research Council (MRC)
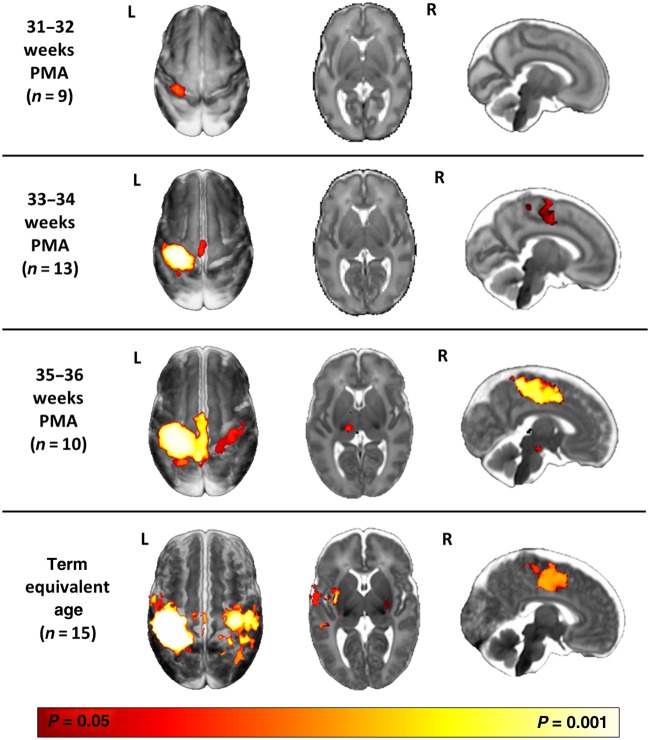
Sensori-motor system Development
The preterm period (equivalent to the third trimester of human gestation) is a key period for the establishment of the sensori-motor system in humans. Working with Professor David Edwards (King’s College London) and Professor Etienne Burdet (Human Robotics Group, Imperial College London) we have developed and used a number of MR compatible robotic devices to systematically study functional responses across the preterm period and following brain injury. Characterising these processes will be crucial to understanding the pathophysiology that underlies cerebral palsy in children.
A review of our methods can be found here:
https://www.frontiersin.org/articles/10.3389/fneur.2014.00197/full

Examples include:
- fMRI responses to hand movement in preterm and term infants:
https://www.sciencedirect.com/science/article/pii/S1053811909011094 - Characterisation of the neonatal HRF following somatosensory stimulation:
https://www.sciencedirect.com/science/article/pii/S1053811912006702 - fMRI responses to olfactory stimuli:
https://onlinelibrary.wiley.com/doi/full/10.1111/apa.12327 - A novel MR compatible device for controlled stimulation of the wrist:
https://link.springer.com/article/10.1007/s10439-013-0782-x - Characterisation of altered connectivity following focal brain injury in preterm infants:
https://link.springer.com/article/10.1007/s00234-014-1412-5 - Systematic study of the evolution of sensori-motor responses across the preterm period:
https://academic.oup.com/cercor/article/26/1/402/2367359 - Characterisation of the sensorimotor homunculus in preterm infants:
https://academic.oup.com/cercor/article/28/7/2507/4975478 - Development of a novel MR compatible device for measuring grasp in infants: https://www.mdpi.com/1424-8220/20/21/6040/htm
- Development of functional connectivity within the sensorimotor cortices across the preterm period: https://onlinelibrary.wiley.com/doi/10.1002/hbm.25785
Excitatory-Inhibitory imbalance in infants at risk of Neurodevelopmetal disorders
Animal models suggest that an imbalance between excitatory and inhibitory neural activity underlies neurodevelopmental disorders such as autism. The aim of this project is to understand if the balance between excitatory (glutamate) and inhibitory (Gamma Amino-Butyric Acid (GABA)) neurotransmitter levels is altered in newborn infants who have a high familial risk of developing neurodevelopmental disorders. To achieve this, we are working with Dr Enrico de Vita and Professor Grainne McAlonan and will use Magnetic Resonance Spectroscopy (MRS) at both 3T and ultra high-field 7T to compare GABA and glutamate levels between groups of control and high risk infants. This new knowledge may help to identify which infants will develop difficulties later in childhood and provide a crucial foundation onto which new therapeutic strategies can be identified and tested.
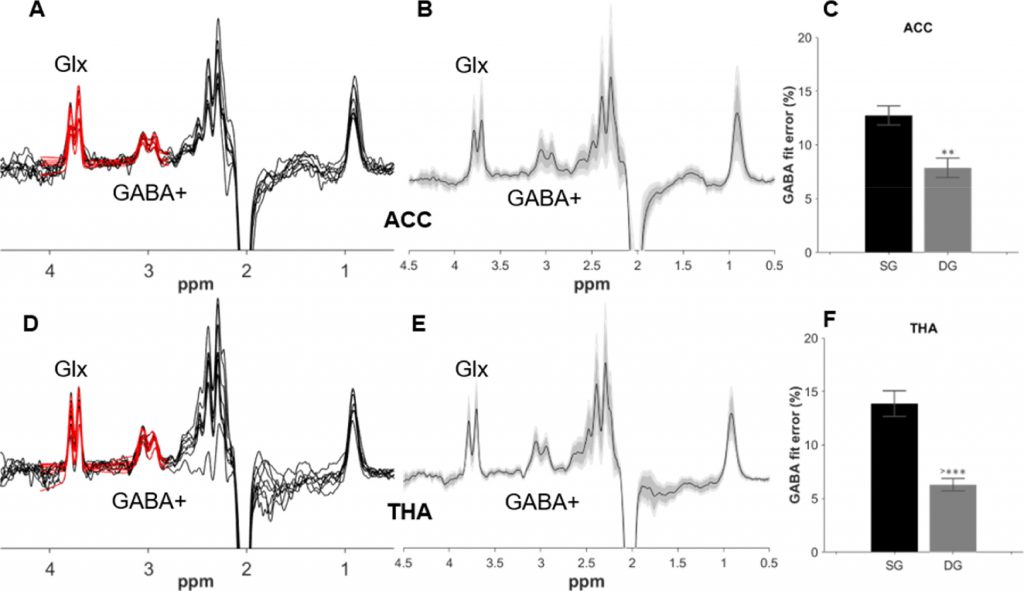
Neonatal GABA+ and Glx edited spectra from the anterior cingulate cortex (A) and thalamus (D) are plotted in black, with double Gaussian fitted model in red. Corresponding group averages from the ACC (B) and thalamus (E) in black, with the standard deviation in dark grey and the 95% confidence interval in light grey. GABA+% fit error (C, F) showing the difference in GABA+ fitting between single Gaussian (SG) and the double Gaussian (DG) models. (Figure reproduced from Yanez Lopez et al. Neuroimage 2021)
A paper describing the optimised acquisition and analysis pipline developed for applying the HERMES sequence with neonates has been published in Neuroimage:
Yanez Lopez et al. Simultaneous quantification of GABA, Glx and GSH in the neonatal human brain using magnetic resonance spectroscopy. Neuroimage 2021; 233: 117930
We were also involved in a review article about edited MRS methods in neonates together with several groups across the UK and USA:
Song et al. Edited Magnetic Resonance Spectroscopy in the neonatal brain. Neuroradiology 2021
The data associated with this manuscript can be provided upon completion of a user agreement, please contact us via email: tomoki.arichi@kcl.ac.uk or enrico.devita@kcl.ac.uk
A systematic review of MRS studies in autism has been recently completed by Alice Thomson as part of her PHD:
Thomson et al. Neurometabolite differences in Autism as assessed with Magnetic Resonance Spectroscopy: A systematic review and meta-analysis. Neuoscience and Biobehavioral Reviews 2024
This work was funded by a grant from Action Medical Research. An article summarising the study is available on their website.
An MR compatible Virtual Reality System
The MRI scanner environment is noisy and claustrophobic, making it challenging for many subjects and impeding imaging of vulnerable populations. Traditional interventions to alleviate anxiety (patient education/preparation, simple audio and video entertainment etc.) provide only limited relief. We are aiming to replace the visual scene with appropriate engaging content which could substantially mitigate claustrophobia and to make the visual experience congruent with physical sensations (noise, vibration, table movement) which could further reduce stress by achieving an integrated experience. Although Virtual Reality (VR) technology could provide a means to do this, in typical applications, immersion is strongly enhanced by user control of the perceived environment through tracking head motion, which is clearly undesirable in MRI. We are therefore developing a fully MRI compatible VR system for brain imaging applications, using gaze tracking for interactive control.
A paper describing the design and features of the VR system has been published in Scientific Reports in 2021:
Qian et al. An eye tracking based virtual reality system for use inside magnetic resonance imaging systems. Scientific Reports 2021; 11: 16301
An a paper describing the instant interaction adaptive gaze control used to allow people to interact with the system can be found here:
Instant interaction driven adaptive gaze control interface. Qian K, Arichi T, Edwards AD, Hajnal JV. Scientific Reports 2024; 14: 11661. A video showing the system in action can be seen here
The patent for the system: WO2020217068A1 MRI SCANNER-COMPATIBLE VIRTUAL REALITY SYSTEM
This work is funded by a MRC Senior Clinical Fellowship and by a project grant from Action Medical Research
A blogpost from Action Medical Research about the project using the system to understand brain processing can be found here
Neural correlates of infant learning
Infants are exposed to many different types of sensory information from very early in development. Animal studies suggest that such patterns of environmental stimulation may play a key role in establishing and consolidating patterns of functional connectivity. However, to encode the complexity of their environment, newborn babies must be capable of processing multi-modal sensory information so that they can interpret the relationship between different stimulus types. Working with Professor Bill Fifer (Columbia University) and using custom engineering techniques we are studying whether neonates can learn an association between two simple sensory stimuli.
The results of this work confirming that associative learning from external sensory experience can shape activity in the developing cortex has been published in Cerebral Cortex:
This work was funded by a Clinician Scientist Fellowship from the Medical Research Council (MRC)
Brain Development in Down Syndrome
Down Syndrome is the most common genetic developmental disorder in humans. It is associated with varying degrees of intellectual disability and delays in speech, memory and learning. As both length and quality of life are improving for individuals with Down Syndrome, it is vital that we can understand the biological substrates of the associated cognitive difficulties so that we can hopefully develop treatments in the future. We are working with Dr Ana Baburamani andProfessor Mary Rutherford to acquire fetal and neonatal MR images from individuals with a diagnosis of Down Syndrome.
A review of this work and the associated video podcast:
https://onlinelibrary.wiley.com/doi/full/10.1111/dmcn.14260,
https://www.youtube.com/watch?v=x4x4iaP7ozk
This work is funded by a grant from Great Ormond Street Sparks.
MAVEHA – Movement Assessment for Very Early Health Assessment
Babies make frequent but seemingly non goal-orientated spontaneous movements throughout the day. Although evidence suggests that with training, it is possible to identify which babies are at high risk of developing cerebral palsy by carefully watching these movements, this process has never been automated. Detailed measures of the movements themselves (such as how fast or smooth the movements are or which parts of the body are involved) and how this changes during the first few months have also never been done. Working with colleagues at Imperial College London led by Dr Bernhard Kainz in the department of computing, we are aiming to combine precise motion tracking methods (3D video and electromagnetic tracking) with machine learning methods to develop a new method which can automatically assess a baby’s movements and help predict which babies may have difficulties later in life.
A paper presented at CVPR 2021 describing a novel deep learning based method to estimate pose in infants from video data is available here:
Schmidtke et al. Unsupervised Human Pose Estimation through Transforming Shape Templates Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR), 2021, pp. 2484-2494
This work was funded by a grant from the EPSRC



